Medical AI · Diagnostics · 2026 Guide
AI in Medical Diagnostics: The Most Accurate Early Detection Tools Used in Hospitals in 2026
aitrendblend.com
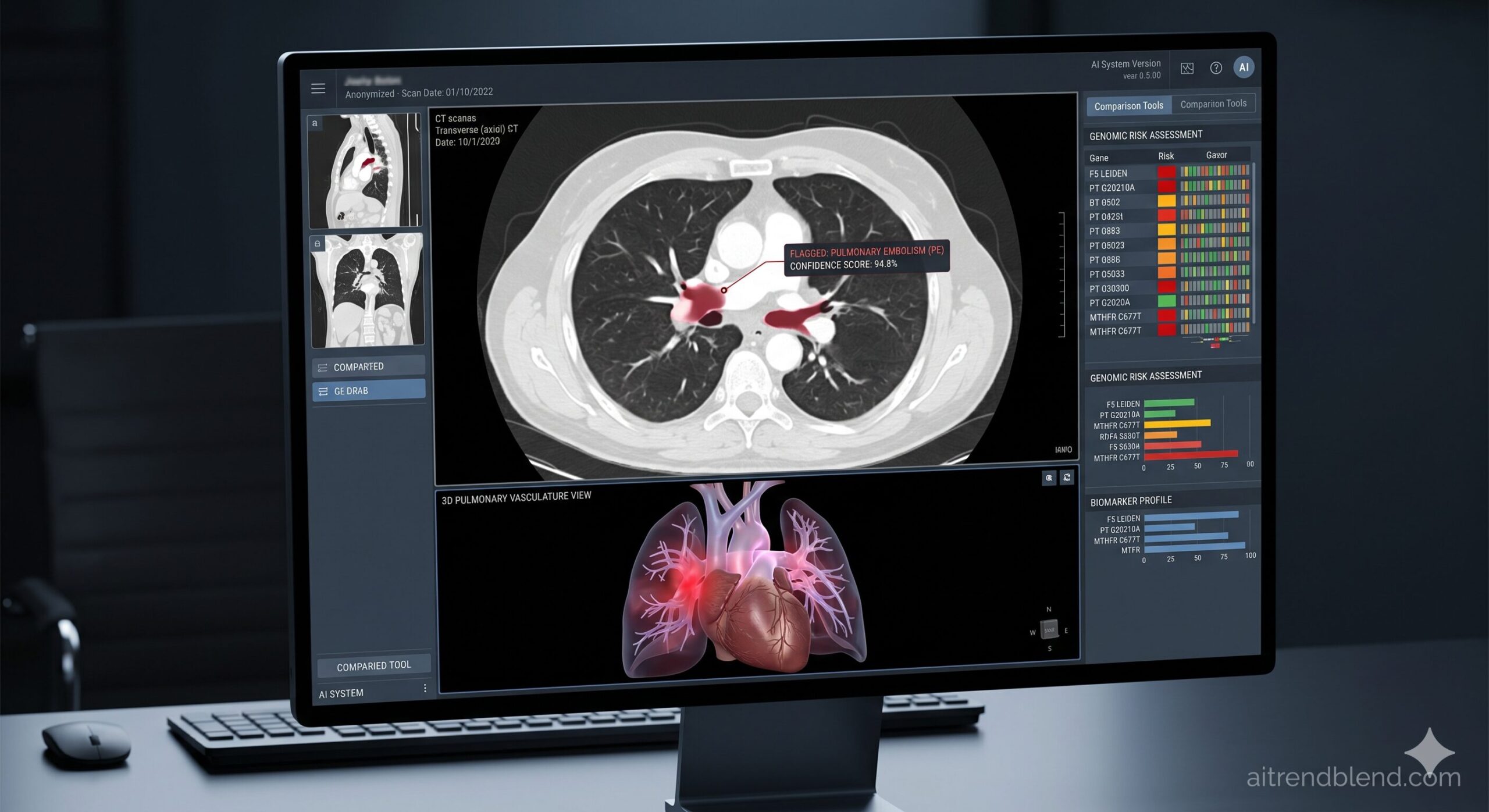
Dr. Amara Osei has reviewed chest CT scans for eleven years. On a Tuesday afternoon shift, she clears a scan — the lungs look clean on visual inspection. An Aidoc alert fires four minutes later: a 6mm pulmonary embolism in the right lower segmental artery, borderline detectable, flagged by the triage AI. She pulls the scan back. It is there. She would have caught it on a second pass. Maybe. That “maybe” is what the AI diagnostics argument actually rests on — not whether the machine is smarter, but whether the combination of human clinical judgment and algorithmic pattern recognition catches more than either does alone.
Medical AI moved from research labs to operating clinical environments at scale between 2022 and 2026. There are now over 950 FDA-cleared or authorised AI and machine learning-based medical devices in the United States — up from fewer than 100 in 2019. The majority of them are diagnostic tools: systems that analyse radiology images, pathology slides, cardiac data, genomic profiles, and retinal photographs to catch disease earlier, triage more accurately, and reduce the cognitive load on clinicians who are increasingly asked to do more with less attention per patient.
This guide covers the ten most consequential AI diagnostic tools deployed in clinical settings as of 2026. Not research prototypes. Not demo videos. Systems that are running in real hospitals, producing real outputs that clinicians are acting on — along with the clinical evidence, FDA status, and real-world performance data that tells you whether to believe the marketing.
One important framing before we begin: all ten tools described here are designed as clinical decision support — they assist physicians, they do not replace them. That is both a regulatory requirement and a clinical necessity. Understanding what that means in practice is the difference between deploying these tools effectively and deploying them irresponsibly.
Why AI Diagnostics Is Different From Every Other AI Application
The stakes in medical diagnostics make the evaluation framework fundamentally different from any other AI deployment category. When an AI writing tool generates a mediocre sentence, you rewrite it. When an AI diagnostic system produces a false negative on an early-stage cancer, a patient may not receive treatment until the disease has progressed past the window where intervention makes the most difference. That asymmetry shapes everything — the regulatory requirements, the clinical validation standards, the deployment protocols, and the ethical obligations of everyone involved in bringing these systems into use.
The metrics matter more here than anywhere else. Sensitivity — the proportion of actual disease cases the system correctly identifies — and specificity — the proportion of healthy cases it correctly clears — are not interchangeable, and the trade-off between them has real clinical consequences. A system optimised for maximum sensitivity will flag more borderline cases, reducing missed diagnoses at the cost of more false positives, more unnecessary follow-up procedures, more patient anxiety, and more radiologist time spent reviewing flagged negatives. A system optimised for specificity reduces the noise burden but risks missing edge cases. Understanding which direction a tool is calibrated for, and why, is essential for deploying it appropriately.
The honest comparison to other medical AI markets: the imaging AI space — radiology, pathology, ophthalmology — is the most mature and evidence-rich. These tools have been tested in prospective clinical trials, published in peer-reviewed journals, and subjected to FDA review. The genomics and clinical decision support space is less standardised; performance claims vary more widely and often rest on retrospective data or smaller cohorts. That distinction shapes how much weight to put on vendor claims in each category.
Key Takeaway
FDA clearance is a floor, not a ceiling. It means the tool met a regulatory bar for safety and substantial equivalence to a predicate device — it does not guarantee the tool performs better than your current standard of care in your specific patient population, on your specific imaging equipment, with your specific patient demographics. Demand institution-level validation data before clinical deployment.
Before You Evaluate: Understanding FDA Clearance and Clinical Evidence
Not all FDA designations are equal, and the distinction matters when evaluating diagnostic AI tools. Most imaging AI receives 510(k) clearance — meaning the FDA determined it is substantially equivalent to a legally marketed predicate device. This is the lowest regulatory bar and does not require clinical trial evidence of superiority. De Novo authorisation applies to novel devices with no clear predicate; it involves more scrutiny. Premarket Approval (PMA) is the most rigorous pathway, reserved for high-risk devices, and requires clinical evidence of safety and effectiveness from prospective studies. Most of the tools in this guide received 510(k) clearance; HeartFlow FFRCT is a notable exception with De Novo authorisation backed by prospective trial data.
When evaluating clinical evidence, apply the same hierarchy you would for any medical literature. Prospective randomised trials with large, diverse patient populations rank above retrospective studies on curated datasets. Published peer-reviewed results rank above conference presentations or white papers. Independent validation on external datasets — not just the dataset the tool was trained on — is the gold standard. Reader studies comparing physician-only versus physician-plus-AI performance are more relevant than standalone AI performance metrics, because that is the workflow being evaluated, not a competition between human and machine in isolation.
510(k) Clearance — “Substantially equivalent” to predicate device
Evidence required: Safety + substantial equivalence (no RCT required)
Most radiology/pathology AI — Aidoc, Viz.ai, Paige AI
De Novo Authorisation — Novel device, no clear predicate
Evidence required: Clinical data supporting safety & effectiveness
HeartFlow FFRCT, IDx-DR (first autonomous AI diagnostic)
PMA (Premarket Approval) — High-risk Class III devices
Evidence required: Prospective clinical trials, highest bar
Rare in diagnostic AI — typically implantable or therapeutic devices
CE Mark (EU) follows different pathways — MDR 2017/745 classification applies.
The question is not whether AI can read a chest X-ray better than a radiologist. In controlled studies on specific tasks, it often can. The real question is whether it makes the entire diagnostic system — radiologist plus AI — better than either working alone.
— aitrendblend.com editorial
The 10 Most Accurate AI Diagnostic Tools Deployed in 2026
The following tools are grouped by clinical domain. Each profile includes regulatory status, key clinical validation data, real-world deployment scale, and an honest assessment of where performance claims should be interrogated. Sensitivity and specificity figures are drawn from peer-reviewed published studies or FDA submissions; real-world performance in uncontrolled settings may vary.
Tool 1: IDx-DR (Digital Diagnostics) — Autonomous Diabetic Retinopathy Screening
IDx-DR holds a singular distinction: it was the first FDA-authorised device to make a clinical screening decision without a physician needing to interpret the result. Approved via De Novo in 2018, it analyses retinal photographs taken by a non-specialist operator and returns one of two outputs — “More than mild diabetic retinopathy detected: refer to an eye care professional” or “Negative for more than mild diabetic retinopathy: rescreen in 12 months.” The physician does not review the image. The AI is the decision-maker.
That is a genuinely different category from every other tool in this guide. It means IDx-DR has been through a more rigorous regulatory bar — De Novo authorisation — precisely because the liability model is different when there is no human physician in the interpretation loop.
Ophthalmology · Autonomous Screening
IDx-DR (Digital Diagnostics)
Designed for primary care clinics where ophthalmologists are unavailable. The value proposition is access: diabetic retinopathy is the leading cause of preventable blindness in working-age adults, and the screening gap is enormous in underserved and rural settings where specialist access is limited. IDx-DR brings screening to the point of care — a primary care visit, a pharmacy, a community health centre.
The FDA pivotal study showed 87.2% sensitivity and 90.7% specificity for detecting more than mild diabetic retinopathy, with an image gradability rate of 96.1%. The 3.9% ungradable rate — images the system cannot evaluate due to quality — are referred to a specialist automatically.
Clinical Verdict
Strongest real-world case for autonomous diagnostic AI in low-resource settings. The access argument is compelling and the validation robust. Sensitivity is lower than expert ophthalmologist review but meaningfully higher than no screening at all — which is the relevant comparison for the populations it is designed to serve.
Tool 2: Aidoc — AI Triage for Radiology at Scale
Aidoc is the closest thing to an industry standard in radiology AI triage. Deployed in over 1,200 hospitals and health systems globally, it runs continuously in the background of radiology workflows — analysing incoming CT scans in real time and flagging critical findings for immediate radiologist review. The system does not report; it prioritises. When a scan shows intracranial hemorrhage, pulmonary embolism, or aortic dissection, Aidoc pushes an alert up the worklist regardless of where the scan was queued.
Radiology · Critical Finding Triage
Aidoc
The 12 FDA-cleared use cases span intracranial hemorrhage, PE, aortic dissection, cervical fracture, rib fracture, appendicitis, COVID-19 detection, and several more. Each clearance is independent and backed by separate validation studies. The breadth of coverage is part of Aidoc’s value proposition — deploying a single platform covers the highest-acuity conditions across emergency and inpatient imaging.
Published real-world data from Cleveland Clinic showed a statistically significant reduction in time-to-treatment for PE when Aidoc was deployed, driven by worklist reprioritisation getting critical scans in front of a radiologist faster. Peer-reviewed studies have shown 18–24% reductions in door-to-treatment time for stroke-equivalent conditions — not because the AI reads faster, but because it eliminates the case from the standard FIFO queue entirely.
Clinical Verdict
The strongest evidence base in radiology AI triage. The value is not accuracy alone — it is the workflow integration: continuous background analysis, zero radiologist overhead for negative scans, and immediate escalation for positives. The ROI case is straightforward for high-volume emergency radiology departments.
Tool 3: Caption AI (GE HealthCare) — AI-Guided Ultrasound for Non-Experts
Echocardiography is one of the most valuable diagnostic tools in cardiovascular medicine and one of the most operator-dependent. Quality cardiac ultrasound images require extensive training — typically years — to acquire reliably. Caption AI changes that equation by providing real-time on-screen guidance during the scan, telling the operator where to move the probe to capture a diagnostic-quality image. The AI handles the interpretation question — “is this image usable?” — so the operator can focus on physical technique.
Cardiology · Ultrasound Guidance
Caption AI (GE HealthCare)
Published evidence showed that approximately 80% of operators with no prior echocardiography experience could acquire diagnostic-quality images for the standard six views within 10 minutes of training. The use case is point-of-care ultrasound — emergency departments, rural clinics, primary care, and pre-hospital settings where a trained sonographer is unavailable but a basic cardiac assessment is clinically essential.
Following GE HealthCare’s acquisition of Caption Health in 2023, the technology has been integrated into Vscan Air SL, the handheld ultrasound device used across primary care and emergency settings globally. The integration means Caption AI guidance ships as standard capability, not a premium add-on.
Clinical Verdict
Best-in-class for the point-of-care access problem. The value is not diagnostic accuracy of the AI model itself — it is the ability to extend cardiac ultrasound capability to clinical settings and operators that would otherwise have none. A strong use case for rural medicine and resource-limited settings.
Tool 4: Viz.ai — Stroke and Pulmonary Embolism Detection Workflow
Time is brain. The phrase applies to stroke care in a precise, clinical sense: every minute of large vessel occlusion (LVO) stroke without treatment costs an estimated 1.9 million neurons. Viz.ai’s core value proposition is minutes — specifically, the minutes between a CT scan being acquired and a stroke team being mobilised. The AI analyses the scan automatically, detects LVO and other critical vascular findings, and sends an immediate notification to the stroke team’s mobile devices — before the radiologist has finished reading the full study.
Neurology / Vascular · Emergency Triage
Viz.ai
A 2020 study published in the Journal of NeuroInterventional Surgery showed 84% LVO detection sensitivity with Viz.ai-augmented workflow versus 66% with standard workflow — a 27% relative improvement. A separate multi-site study documented a mean time saving of 37 minutes from CT acquisition to stroke team notification. At the scale of 1,400+ hospitals, that time differential translates to measurably better neurological outcomes across thousands of patients per year.
Beyond stroke, Viz.ai has expanded clearances to cover pulmonary embolism, aortic dissection, and other time-critical vascular findings. The platform also includes care coordination tools — the notification system allows direct communication between the radiologist, stroke neurologist, and interventional team via a HIPAA-compliant mobile interface, replacing the chain of phone calls that typically added further delay.
Clinical Verdict
The clinical evidence for stroke workflow improvement is the strongest of any AI diagnostic tool in this guide. The 37-minute time saving is prospective, multi-site validated, and directly linked to patient outcomes data. For comprehensive stroke centres handling significant LVO volume, the case for deployment is robust.
Tool 5: Paige Prostate — The First FDA-Cleared AI Pathology Tool
Paige Prostate earned another historical first: the first FDA-cleared AI for use in anatomic pathology, cleared in 2021. Pathologists reviewing digitised prostate biopsy slides use Paige Prostate as a second reader — the AI analyses the slide independently and flags any areas containing cancer that the pathologist’s initial review might have graded as benign or overlooked entirely.
The critical use case is not replacing the pathologist’s read — it is a safety net for the small but meaningful percentage of cases where cancer is present but subtle enough to downgrade on a primary review. Prostate cancer’s Gleason grading system has well-documented inter-observer variability; AI provides a consistent, calibrated second opinion that does not fatigue.
Pathology · Prostate Cancer Detection
Paige Prostate (Paige AI)
The pivotal FDA study showed 97.8% sensitivity for clinically significant prostate cancer (Gleason ≥6), and a reader study demonstrated that pathologists using Paige Prostate as a second reader reduced missed cancer cases by approximately 70% compared to their unassisted performance. That figure requires careful interpretation — it represents the reduction in cases where cancer was present but the pathologist’s unassisted primary read would have classified the slide as benign. The absolute miss rate for experienced pathologists on well-prepared slides is already low; the AI catches the long tail of the hardest cases.
Paige AI has expanded beyond prostate to breast, skin, and lung pathology, with separate validation programmes under way. The platform integrates with major digital pathology scanners including Philips IntelliSite and Leica Aperio, fitting into existing digital pathology lab workflows without requiring hardware replacement.
Clinical Verdict
The strongest evidence base in digital pathology AI. The 70% reduction in missed clinically significant cancer figure is compelling and the training dataset scale is industry-leading. Primary adoption barrier is the requirement for digital pathology infrastructure — labs still using glass slides need scanner investment before Paige is accessible.
Tool 6: HeartFlow FFRCT — Non-Invasive Coronary Artery Disease Assessment
Coronary artery disease assessment traditionally required an invasive catheterisation procedure — threading a wire into the coronary arteries to measure blood flow restriction directly. HeartFlow FFRCT replaced that with a computation from a standard coronary CT angiogram. The algorithm builds a patient-specific 3D model of the coronary arteries from the CT data, then simulates blood flow using computational fluid dynamics to calculate fractional flow reserve (FFR) — the same physiological measurement obtained invasively — without a catheter.
Cardiology · Coronary Artery Disease
HeartFlow FFRCT
The PLATFORM randomised trial — published in JACC — showed that HeartFlow-guided management led to a 50% reduction in unnecessary invasive coronary angiograms with non-obstructive disease. The patients who did not get catheterised due to HeartFlow guidance had equivalent one-year clinical outcomes to those managed with standard care, validating the safety of the non-invasive pathway.
The clinical and cost implications are significant. Invasive coronary angiography carries procedural risk — stroke, bleeding, contrast nephropathy — and costs roughly $10,000–$15,000 per procedure. Eliminating half of those in diagnostically unclear patients represents both a safety improvement and meaningful cost reduction. HeartFlow is now reimbursed under Medicare and most major commercial payers in the United States.
Clinical Verdict
Strongest clinical and economic evidence in cardiac diagnostic AI. The PLATFORM trial design is rigorous, the outcome data is compelling, and the reimbursement pathway is established. The 79% specificity means roughly 1 in 5 lesions classified as haemodynamically significant will be false positives — an important limitation when the clinical consequence of a positive is invasive intervention.
Tool 7: PathAI — Multi-Cancer Pathology Analysis and Biomarker Quantification
PathAI is building what might be described as the foundational model layer for computational pathology. Where Paige focuses on detection — is cancer present or absent — PathAI’s platform extends into biomarker quantification, tumour microenvironment analysis, and treatment response prediction from standard H&E and immunohistochemistry slides. These are outputs that pathologists could previously only approximate semi-quantitatively; AI makes them reproducible and scalable.
Pathology · Multi-Cancer Biomarker Analysis
PathAI
PathAI’s most clinically immediate applications are in breast cancer (HER2 scoring, PD-L1 quantification), liver disease (NASH fibrosis staging), and lung cancer (tumour mutational burden, MSI status). Each of these biomarkers influences treatment selection — a patient whose tumour is HER2-positive receives targeted therapy; one with MSI-high status is a candidate for immunotherapy. Quantifying these biomarkers reproducibly and at scale is where PathAI’s technology has its highest current clinical impact.
The biopharma partnership model is central to PathAI’s current revenue — AstraZeneca, Bristol Myers Squibb, and Eli Lilly use PathAI’s platform for clinical trial patient selection and treatment response monitoring. This gives the company access to proprietary clinical datasets that strengthen model performance in a virtuous cycle unavailable to academic research groups.
Clinical Verdict
Less applicable at the individual hospital deployment level than Aidoc or Viz.ai — PathAI’s current strength is in biopharma research partnerships and clinical trial support. The biomarker quantification applications are clinically meaningful but many require lab-specific validation before clinical use. Watch this space: the NASH and HCC tools have the most immediate clinical deployment pathway.
Tool 8: Tempus AI — Oncology Genomic Profiling and Treatment Matching
Tempus approaches cancer diagnostics from a different angle than the imaging and pathology tools above: it analyses the molecular profile of a patient’s tumour to identify the specific genetic mutations, fusions, and biomarkers that determine which targeted therapies are most likely to work. The platform combines next-generation sequencing with one of the largest proprietary clinical-genomic databases in the world — over 50 million de-identified clinical records and more than two million genomic profiles — to match tumour profiles to therapies with supporting evidence.
Oncology · Genomic Profiling & Treatment Matching
Tempus AI
Tempus’s xT CDx assay — a comprehensive genomic profiling panel covering 648 genes — received FDA breakthrough device designation. The platform is deployed at over 1,000 institutions and is integrated into oncology workflows at major academic medical centres. Oncologists receive a Tempus report alongside pathology results that includes actionable genomic findings, matched FDA-approved and clinical trial options, and relevant published literature — compressed into a single clinical decision support document.
The unique value is the scale of the clinical-genomic database. When a patient presents with a rare KRAS G12C mutation in a pancreatic cancer, Tempus can draw on real-world outcome data from hundreds of patients with the same mutation across its partner network — data that no single institution could accumulate alone. That evidence layer distinguishes Tempus from general-purpose genomics labs running the same sequencing panels.
Clinical Verdict
The leading comprehensive genomic profiling platform in oncology for real-world clinical use. The database scale is a genuine competitive moat. Adoption is high at academic medical centres; community oncology is the growth frontier. The caveat: turnaround time for full genomic profiling is typically 10–14 days, which makes urgency of clinical need an important factor in sequencing order timing.
Tool 9: Med-Gemini (Google DeepMind) — Multimodal Clinical Reasoning
Med-Gemini represents a different architecture from every other tool in this guide. It is not a narrow specialist model trained on one imaging modality — it is a multimodal foundation model fine-tuned on medical data, capable of reasoning across text, images, audio, and structured clinical data simultaneously. The practical question is whether that generality translates to clinical usefulness in the specific, high-stakes context of medical diagnosis — and the answer in 2026 is: on benchmarks, impressively; in deployment, with important caveats.
Clinical AI · Multimodal Foundation Model
Med-Gemini (Google DeepMind)
Published benchmarks are striking. Med-Gemini Ultra achieves over 90% on the USMLE Step 1-3 clinical knowledge exams and 91.1% on MedQA — surpassing the published performance of GPT-4, Med-PaLM 2, and earlier Gemini variants. Multimodal evaluations showed strong performance on dermatology image classification, chest X-ray findings, and ECG interpretation when combined with clinical history.
The deployment picture is more complex. Med-Gemini is not yet available as a general-purpose clinical tool outside of structured research partnerships and Google Cloud’s Vertex AI platform for healthcare organisations. The path from benchmark performance to FDA-cleared clinical deployment involves prospective clinical validation, regulatory submission, and integration into certified EHR systems — a process that takes years even for organisations with Google’s resources. What Med-Gemini represents in 2026 is the most capable general-purpose medical reasoning engine available, operating in research and pilot contexts while the regulatory pathway matures.
Clinical Verdict
Best benchmark performance of any AI system on standardised medical knowledge tests. The clinical deployment gap — between benchmark excellence and FDA-cleared production use — remains wide. The most immediate real-world applications are clinical documentation, literature synthesis, and structured data extraction from EHRs where the regulatory bar is lower than direct diagnostic use.
Tool 10: The Integrated Early Cancer Detection Pipeline
This is not a single product — it is the architecture that leading academic cancer centres are deploying in 2026, combining multiple AI modalities into a unified early detection system. The sum of its parts outperforms any individual tool because different modalities catch different cancers at different stages. Imaging AI catches structural abnormalities. Genomic profiling identifies hereditary risk and actionable mutations. Liquid biopsy detects circulating tumour DNA before a mass is large enough to image. Clinical NLP extracts relevant history from unstructured notes. Combining these streams changes what early detection means in practice.
Why It Works: Each layer catches what the others miss. Liquid biopsy detects methylation signatures before imaging can resolve a mass. Imaging AI catches the structural lesion that liquid biopsy may not have sufficient signal for at early stage. Genomic profiling determines whether the confirmed cancer requires targeted therapy or standard chemotherapy. No single layer is sufficient; the combination is the advancement.
How to Adapt It: For community hospitals without full academic cancer centre infrastructure, the highest-value starting point is Layer 1 (risk stratification from EHR data, available through Epic and Cerner modules) plus Layer 2 (imaging AI, deployable without hardware changes). Those two layers alone close meaningful gaps in the catch rate for the highest-volume cancers.
Common Misconceptions That Lead to Poor Deployment Decisions
The failure modes in medical AI deployment differ from other sectors because the consequences of getting it wrong are not correctable with a quick update. A missed diagnosis that AI should have caught is a different kind of failure than a misclassified email. Understanding the specific ways that AI diagnostic tools underperform — and why — is as important as understanding how they perform at their best.
The most damaging misconception is that FDA clearance certifies clinical superiority. It does not. It certifies that the tool met a regulatory standard for safety and substantial equivalence — typically to an existing predicate device. A cleared AI tool may perform worse than the standard of care at your specific institution for your specific patient population, and the clearance documentation would not tell you that. Demand validation data from your institution type, your scanner model, and your patient demographics before deployment decisions.
Key Takeaway
Sensitivity and specificity are trade-offs, not independent dials. A tool optimised for maximum sensitivity — catching every disease case — will generate more false positives, increasing radiologist workload and patient anxiety. Before deploying, understand which direction the tool is calibrated and what that means for your workflow. A tool calibrated for high sensitivity in a high-volume screening programme creates different workflow demands than one calibrated for high specificity in a tertiary referral centre.
Viz.ai LVO Stroke: Sensitivity 84% · Specificity ~90%
Aidoc ICH (CT): Sensitivity ~91% · Specificity ~89%
Paige Prostate: Sensitivity 97.8% · Specificity ~72%
IDx-DR Retinopathy: Sensitivity 87.2% · Specificity 90.7%
HeartFlow FFRCT: Sensitivity 86% · Specificity 79%
Galleri Liquid Biopsy: Sensitivity ~66% (Stage I–II) · Specificity 99.5%
Figures from published FDA submissions and peer-reviewed studies. Real-world performance may vary.
| Misconception | Wrong Assumption | Clinical Reality |
|---|---|---|
| FDA clearance = proven accuracy | “FDA cleared means it outperforms our current radiologists” | Clearance means substantial equivalence to a predicate device — not superiority. Demand institution-specific validation data |
| Trial performance = real-world performance | “Published sensitivity of 91% will hold in our department” | Curated trial datasets often outperform real-world deployment due to scanner differences, protocol variation, and patient population shifts |
| Demographic universality | “A tool validated on diverse trial populations works equally well across all our patients” | Most AI diagnostic tools show measurable performance gaps across race, age, BMI, and imaging equipment. Audit output equity during deployment |
| AI as final decision-maker | “If Aidoc clears the scan, we don’t need to review it” | All FDA-cleared diagnostic AIs are designed as clinical decision support — the physician retains clinical and legal responsibility for the diagnostic conclusion |
| Alert fatigue is someone else’s problem | “More AI alerts means more catches — that’s always better” | Poorly calibrated tools with low specificity generate alert fatigue that leads radiologists to override AI flags — including correct ones. Specificity matters as much as sensitivity |
What AI Medical Diagnostics Still Cannot Do in 2026
The evidence base for AI in medical diagnostics is genuine and growing, but the gap between benchmark performance and clinical deployment at scale contains real limitations that the field has not yet solved. The most important is demographic performance disparity. Studies across multiple imaging AI systems have documented that sensitivity and specificity can vary by 5–15 percentage points across racial and ethnic groups, age ranges, and body types — typically with worse performance in populations underrepresented in training data. An AI system that catches 91% of intracranial hemorrhages in a trial population drawn predominantly from academic medical centre patients may perform at 80–85% in a community hospital serving a different demographic. This is not a hypothetical concern — it has been documented in dermatology AI (where lighter skin tones consistently outperform darker tones on published lesion classification tools), in chest X-ray AI, and in certain ECG analysis tools. The FDA has issued guidance on algorithmic transparency and bias evaluation; deployment without institution-level equity auditing remains an ethical gap.
A second genuine limitation: distribution shift. AI diagnostic models are trained on data from a specific distribution — scanner models, imaging protocols, patient populations, disease prevalences. When the deployment environment differs from the training distribution, performance degrades in ways that can be difficult to detect without deliberate monitoring. A radiology AI trained on Siemens CT data may perform differently on GE equipment running different reconstruction algorithms; a model trained on 2022–2024 data may underperform on a disease presentation shifted by a new dominant variant or treatment pattern. Ongoing performance monitoring after deployment is not optional; it is the mechanism by which distribution shift is detected before it causes clinical harm.
Finally, the problem that all ten tools in this guide navigate carefully: clinical context. AI diagnostic systems process the data they are given — the imaging file, the genomic sequence, the retinal photograph. They do not know that the patient’s family cannot afford the imaging follow-up a positive result would trigger, or that the patient is in palliative care and a new cancer diagnosis would change nothing about management, or that the “abnormality” flagged on the chest CT is a known post-surgical change documented in a note the AI did not read. Clinical context transforms raw diagnostic data into clinical decisions. The physicians who use these tools are not simply interpreters of AI output — they are the layer that makes AI diagnostic information actionable, safe, and appropriate for the specific patient in front of them.
What Has Actually Changed in Early Detection
The ten tools in this guide collectively represent a genuine advance in what early detection means in clinical practice. Not because any individual tool is perfect — none of them is — but because the floor has been raised for the most consequential diagnostic tasks. A pulmonary embolism that might have sat in a radiology queue for 90 minutes now triggers an alert in under five minutes. A prostate biopsy slide that might have been cleared on a fatigued afternoon read now gets a second set of algorithmic eyes calibrated on a million training examples. A patient at high genomic risk for breast cancer in a community practice now has access to the same molecular profiling that elite cancer centres have offered for a decade. Those are real changes, with real patient consequences, happening at scale.
What this means for the practice of medicine is subtler than the marketing suggests. AI did not replace the radiologist or the pathologist. What it did was change the question they are asked. Before AI triage, the question was: “of the 80 scans in my queue, which ones have a finding?” Now it is: “the AI flagged these three as critical — do I agree, and what should happen next?” That is a narrower, faster cognitive task — and a different one, requiring different judgment. Learning to work well with AI diagnostic tools is as much a clinical skill as learning to read an ECG, and it is one that medical education has only recently begun to take seriously.
The equity dimension of medical AI is the one that deserves the most scrutiny from anyone evaluating these tools for deployment. The risk is not just that AI is imperfect — all diagnostic tools are imperfect. The risk is that AI imperfections are systematically worse for already-underserved populations, and that deploying unaudited tools at scale could widen the diagnostic equity gap rather than close it. The tools that take this seriously — with transparent bias testing, demographic performance data in their clearance submissions, and ongoing post-market monitoring protocols — are the ones worth deploying. Those that do not are worth interrogating before you bring them into clinical use.
Over the next 12 to 18 months, the most consequential developments will likely come from two directions: liquid biopsy at scale, where Grail’s Galleri and competitors are pushing cancer signal detection sensitivity upward in prospective screening trials; and multimodal clinical reasoning, where Med-Gemini-class models are advancing toward the kind of holistic patient-level analysis that no single-modality specialist AI can provide. The integration of imaging, genomics, liquid biopsy, and clinical history into a unified early detection pipeline is already the architecture at leading cancer centres. It will become accessible infrastructure within 24 months. When it does, the definition of “early detection” will shift further than it has in any previous decade.
Stay Ahead of Medical AI Developments
The medical AI space moves faster than any institutional procurement cycle. Our monthly digest tracks new FDA clearances, published clinical validation studies, and deployment evidence as it becomes available.
